Saturday, December 23
I spent the past week listening to Nikhil Kamath's podcast on EVs and learned many interesting details about them, especially how they use the same lithium-ion batteries as our cell phones. One common concern about EVs is the battery life, and it's similar to what happens with our phone batteries over time. Have you noticed that as the years go by, your phone's battery seems to drain more quickly? Well, that's something we can't avoid. The only thing we can control is how long it takes for the lithium-ion battery in your phone or EV to wear out to the point where you need to replace it.
Elon Musk recently explained how lithium-ion batteries work in simple terms on The Joe Rogan Experience. Imagine a big empty parking lot. As cars arrive, they look for spots to park. Initially, with plenty of space, it's easy for cars to find a spot. However, as the lot fills up, it becomes harder for them to find an empty space. Think of the parking space as the liquid in the battery and the cars as ions.
Now, let's extend the analogy a bit. Since these cars are driven by humans, they behave like humans. Just as a lack of opportunities can be a problem, too many opportunities also bring issues. When cars arrive initially, they get confused about where to park since there's a lot of space (0-20%). As the lot fills up a bit (20%-80%), it becomes easier for others to find space, as they see the orientation and space is not too filled or too empty. However, once it crosses 80%, it becomes a problem again for ions to find parking space.
For optimized battery life, it's suggested to keep your phone between 20 percent and 80 percent. While seeing 100 percent charge might be reassuring, it's not ideal for the battery. A lithium-ion battery doesn’t like to be fully charged.
But how do the batteries actually operate?
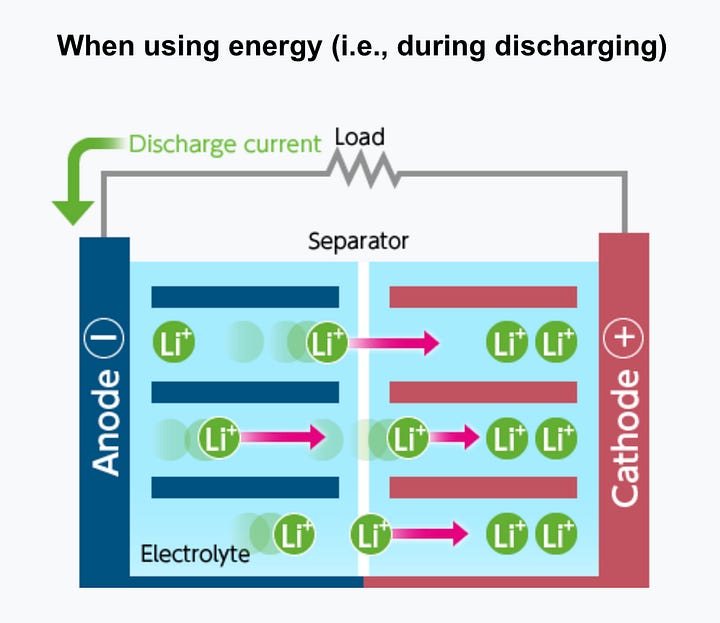
A rechargeable lithium-ion battery, like the one in your phone, is made up of parts called cells. Each cell has three main things: a positive part (connected to the plus sign), a negative part (connected to the minus sign), and something called an electrolyte in between. The positive part is usually made of a chemical called lithium-cobalt oxide or, in newer batteries, lithium iron phosphate. The negative part is typically made of carbon (graphite).
Here's the basic idea of how it works: When you charge the battery, the positive part gives up some of its lithium ions. These ions move through the electrolyte to the negative part and stay there, storing energy. When you use the battery (discharge), the lithium ions move back to the positive part, creating the energy that powers your device. In both cases, electrons move in the opposite direction around the outer circuit.
The movement of ions through the electrolyte and electrons around the circuit are connected processes. If one stops, the other stops too. For example, if the battery completely runs out, ions can't move, and you lose power. Also, if you turn off what the battery is powering, the flow of electrons stops, and so does the flow of ions. The battery slows down discharging when disconnected, but it still happens slowly.
Unlike simpler batteries, lithium-ion ones have controllers to manage how they charge and discharge. These controllers prevent problems like overcharging and overheating, which could make the battery explode in some situations.

Having understood how the battery works, consider your battery’s overall lifespan as a resource you’ll need to spend wisely to preserve your own sanity. It’s up to you to decide what safeguards are worth the trouble.
See you next weekend, have a wonderful week ahead :)
Cheers!
The future sounds amazing. Last week I tried a new AI tool called as Suno AI. This tool will help you in transforming any lyrics into a song of your type.
Incredible stuff. Type in your lyrics, choose the music style and voila, you are done!
Check out some samples that I created -








